
In this article, I will provide an overview of the various technical audit techniques and the technical requirements covered in three types of technical audits. The technical auditors must clearly communicate with auditees that gaps will be identified together in order to drive improvement, not to find “fault” and cast blame. Management should encourage an open, fair, collaborative effort between technical auditors and auditees. This involves auditing both management and technical activities to identify gaps, and to drive improvements.Ī positive audit culture is essential to ensure that internal technical audits are effective. However, if you prefer to have your own internal audit checklist, then you may want to consider this checklist.Laboratories are required to have a program of internal audits to assess whether their operations comply with ISO/IEC 17025 requirements on an ongoing basis. Or, you may be able to log into your account or portal to download a copy. 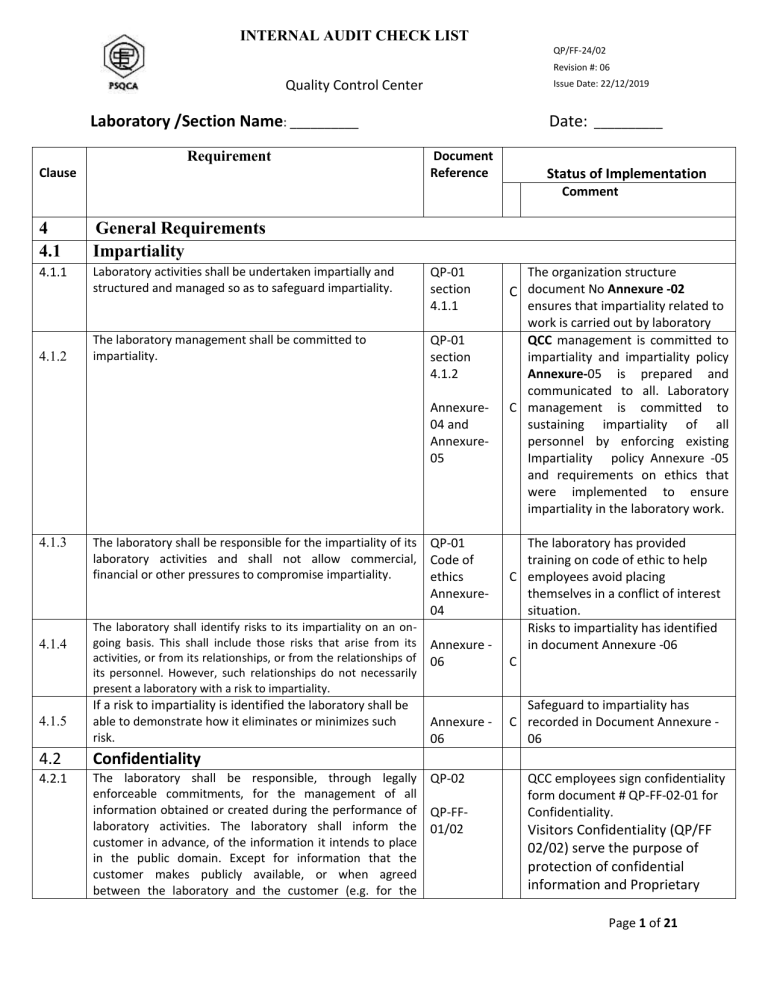
You should already have a checklist, or you can always use the checklist that your accreditation body uses. If your laboratory is already ISO/IEC 17025:2017 accredited, you may not need this checklist. That is why I recommend this ISO/IEC 17025 internal audit checklist to you. 
Fixing deficiencies after an audit requires a lot more documentation and objective evidence (for you to send to the accreditation body) which will cause you to spend more time and money. It is best for you to find problems with your management system before an assessor does. Help you learn more about your quality management system.Give you experience performing internal audits, and.Reduce the number of deficiencies assessors will find,.Find gaps in your quality management system,.If you are a laboratory that is applying for ISO/IEC 17025:2017 accreditation, I recommend that you perform an internal audit before your assessment. Make sure to pay extra attention to these requirements to ensure that your quality management system meets requirements. When you see a yellow, highlighted section, you know that this requirement is one of the most common deficiencies cited during ISO/IEC 17025 assessments. 
When you see an asterisk (*), make sure that you review records or other objective evidence that support the quality management system and meets ISO/IEC 17025:2017 requirements. When you see a thick, black border, make sure the quality management system has a documented procedure, process, plan, criteria, or other relative document. Therefore, I made this checklist to help you out. Especially, if you are a laboratory that is applying for accreditation. However, this is not always available to everyone. Truthfully, I like to conduct internal audits using the same assessment checklists used by accreditation bodies. Highlight the most common deficiencies cited during ISO 17025 assessments.Indicate when you need to review objective evidence (e.g.Show you when a procedure, process, or document is needed,.ISO/IEC 17025:2017) in it, because you should have a copy of it. It does not include the written standard (i.e. This 22-page audit checklist was designed using my favorite elements from the same accreditation body checklists that assessor’s using to audit your laboratory. If so, this ISO/IEC 17025 Internal Audit Checklist is for you. The Best ISO/IEC 17025 Internal Audit Checklist Availableĭo you need to perform internal audits for ISO/IEC 17025:2017 accreditation?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
